Choose language
Choose language
< Return to main menu
 Choose language
Choose language


a biotech company in the US
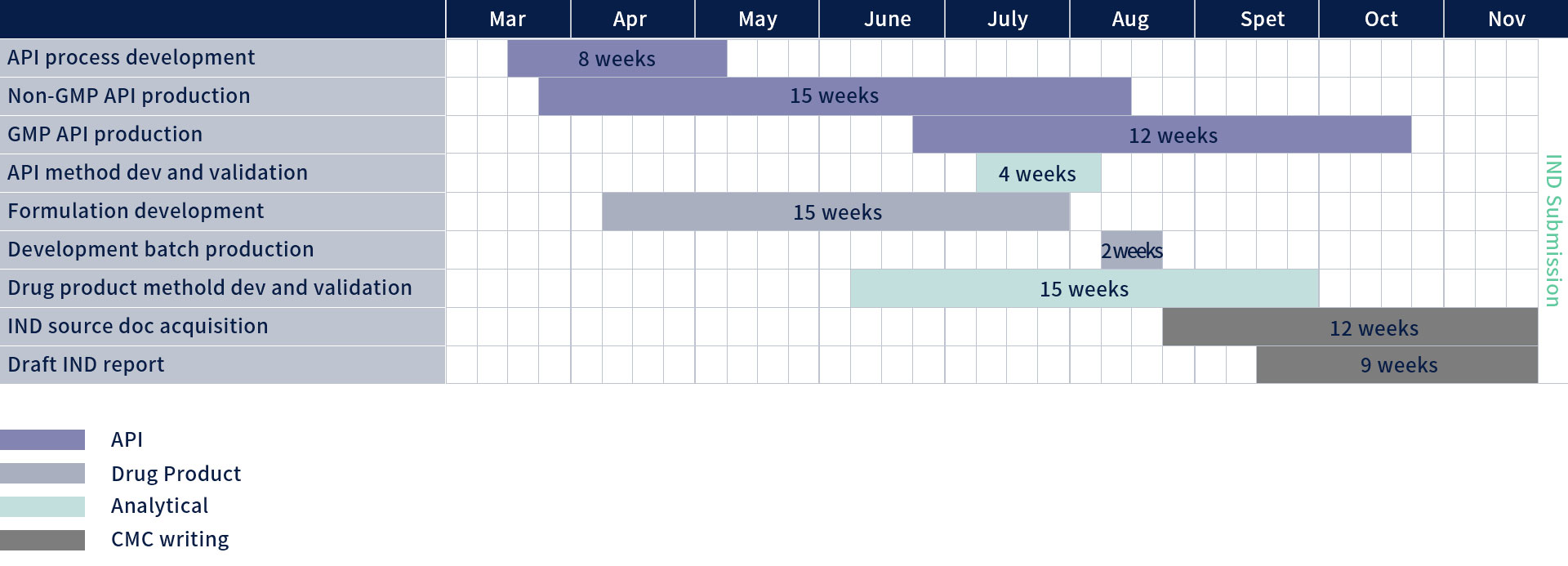
API process development and manufacturing
Formulation development and manufacturing
CMC writing for IND filing
Extremely tight timeline: tox study in 4 months and IND filing in 10 months
Technical challenge: low yield from original route
Supply challenge: very limited amount of raw material
Developed a new route within 1month; reduced reaction step from 30 to 25; purity increased to 50%; overall yield is 4 times higher
With only 5 gram of API, developed analytical method for 4 weeks
With only 7 gram of API, Injectable formulation for manufacturing was developed in just 2 days
Completed the DS IND Module 3 dossier 4 weeks after GMP API batch was released